What is nitrogen?
Nitrogen (N) is one of the main elements that make up living organisms, along with carbon, oxygen and hydrogen. On average, it accounts for 2.5 to 3.5% of the dry organic matter in animals. Amino acids, which are the building blocks of proteins, are all composed of an amine group (-NH2) containing nitrogen and a carboxylic acid group (-COOH). Nitrogen is therefore the third most abundant element in amino acids, after carbon (50-55%) and oxygen (20-23%). Nitrogen is therefore essential for ensuring proper metabolism, protein synthesis and, consequently, animal growth.
1.Protein requirements, metabolism in aquaculture and nitrogen emissions
25 to 40%
These are the average dietary protein requirements of aquaculture species.
Aquaculture species generally have higher dietary protein requirements than other farm animals such as poultry (15–22%) (Applegate and Angel, 2014). These higher amino acid requirements are explained by the fact that amino acid oxidation is the primary source of energy in fish (Weber and Haman, 1996).The dietary protein requirements of fish vary according to species and diet, ranging from around 25-35% for omnivorous and herbivorous species such as tilapia and carp, to 35-40% for carnivorous species such as salmonids, sea bass and sea bream.
Proteins ingested by animals are rapidly broken down into amino acids in the digestive tract by the action of various specific enzymes: pepsins first break down proteins into peptides, then trypsin and chymotrypsin break down the peptides into amino acids (Engrola et al., 2009; Gamboa-Delgado et al., 2011). These amino acids are then absorbed by intestinal cells and used to synthesise new proteins or to produce energy through deamination (Bröer, 2008). The amine group is converted into ammonia (NH3), a metabolic waste product that is toxic to animals if it accumulates (Forster and Goldstein, 1969). Aquatic organisms can rapidly excrete NH3 into the water through their gills, thus avoiding its conversion and conserving energy compared to urothelial animals such as mammals and birds (Figure 1).
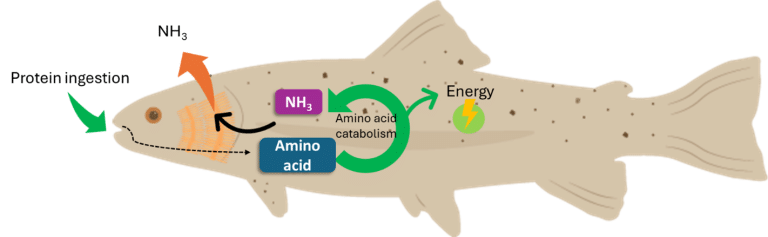
Figure 1: Protein metabolism and nitrogen excretion
2. The nitrogen cycle and its various forms in water
Once released into water, NH3 dilutes easily. Depending on water parameters, mainly pH and temperature, the dominant form will be NH3 or ammonium (NH4+), a form that is less toxic to fish and shrimp, by capturing an H+ ion (Thurston and Russo, 1983). At acidic pH, the dominant form is NH₄⁺, while at basic pH (>8.5), the most prevalent form is NH₃. Temperature also influences the ionisation of NH₃, with higher temperatures favouring the NH₃ form over NH₄⁺.
8.5
This is the pH that must not be exceeded in order to limit the formation of NH3.
It is the biological process that transforms NH3 into NO2- and then NO3- through the action of bacteria.
Next, the biological process of nitrification transforms NH3 into nitrate (NO3–) under the action of microorganisms. Nitrification is divided into two distinct stages: nitrosation and nitration, each carried out by different microorganisms. Nitrosation, carried out by bacteria of the genera Nitrosomonas, Nitrosococcus, and Nitrosospira, converts NH3 into nitrite (NO2–). Nitration, carried out in particular by the genera Nitrobacter, Nitrococcus, and Nitrospira, transforms NO2– into nitrate (NO3–). The oxidation of NH3 and NO2– provides bacteria with the energy they need to convert CO2 into organic matter (Figure 2).
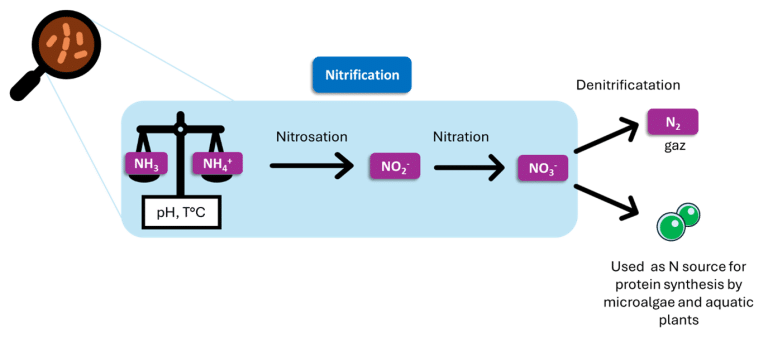
Figure 2: Nitrogen cycle in water
(Robles-Porchas et al., 2020)
To optimize nitrification, it is crucial to maintain neutral pH, good oxygenation, and redox balance in fish and shrimp ponds (Molins-Legua et al., 2006). The NO3– produced by nitrification is then absorbed and used as a source of nitrogen by aquatic plants and algae, which reduce NO3– to NH4+ to synthesize amino acids and proteins. Any disruption of the nitrogen cycle can negatively affect the performance and survival of fish and shrimp by generating toxic nitrogen compounds, reducing the concentration of oxygen (O2), and altering the pH of the water.
- In lake trout (Salvelinus namaycush), exposure to 198 µg/L of ammonia for 60 days reduces protein efficiency by 7% (Beamish and Tandler, 1990).
- Rainbow trout fry (Oncorhynchus mykiss) exposed to 13 mg/L of ammonia for 90 days after hatching saw their weight decrease by 54% and their survival rate by 70% (Brinkman et al., 2009).
- In whiteleg shrimp, the accumulation of nitrite and ammonia from faeces and uneaten food over 33 days reduces survival rates by 2.5 times and growth rates by 20.5% (Han et al., 2017).
- In river catfish (Ictalurus punctatus), exposure to 3.71 mg/L for 31 days reduces growth by 20% (Colt et al., 1981).
3. Harmful effects of nitrogen in water on aquatic animals
Nitrogen released into water can affect aquatic animals directly or indirectly. The different forms of nitrogen described above, including NH3, NH4+, NO2–, and NO3–, have different levels of toxicity for fish and shrimp. While the limit concentrations in water for the various nitrogen compounds vary depending on the species, the order of toxicity remains the same for all species: NH3 is the most toxic, followed by NO2–, then NH4+ and finally NO3– (Figure 3).
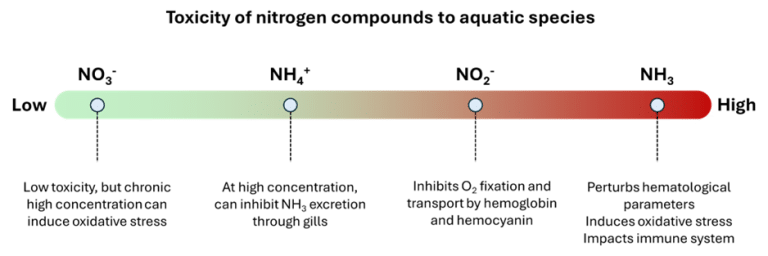
Figure 3: Toxicity of nitrogen compounds to aquatic species (Lin et al., 2023; Tomasso, 1994)
a. Direct toxicity of nitrogen
Since NH3 is an uncharged molecule, it can easily diffuse through biological membranes, particularly gills (Klocke et al., 1972; Randall and Wright, 1987). Once in the blood, it disrupts many hematological parameters, including acid-base balance, coagulation, and molecule transport. In fish, the accumulation of NH3 disrupts the immune system, induces oxidative stress, and causes tissue damage in the gills, liver, kidneys, and intestines. In crustaceans, NH3 also induces oxidative stress and affects the innate immune system and molting (Bouwman et al., 2011; Chang et al., 2015; Cui et al., 2017; Hong et al., 2007; Rodríguez-Ramos et al., 2008; Romano and Zeng, 2013).
- Immunosuppression
- Oxidative stress
- Neurotoxicity
- Gill stress
NO2– is less toxic than NH3, but its toxicity remains very high. In fish, the main mechanism of NO2– toxicity is the formation of methemoglobin (MetHb) through the oxidation of iron (Fe) in hemoglobin. MetHb can no longer bind and transport O₂, causing hypoxia despite oxygenated water (Jensen, 2003). A similar phenomenon is observed in shrimp: NO₂⁻ oxidizes the copper (Cu) in hemocyanin, preventing O₂ binding. NO₂⁻ can also induce oxidative stress and disrupt the innate immune system of fish and shrimp. NO₂⁻ also impacts the biology and health of gills (Svobodová et al., 2005), reduces food intake in fish and shrimp (Ribeiro-de-Campos et al., 2021; Roques et al., 2013) and affects their growth (Ciji and Akhtar, 2020; Colt et al., 1981; Frances et al., 1998; Koo et al., 2005; Mallasen and Valenti, 2006; Siikavuopio and Sæther, 2006).
NH4+ is much less toxic, and its concentrations in water must be very high to affect aquatic animals. At high concentrations, NH4+ can limit the ability of gills to excrete NH3, leading to the accumulation of NH3 in fish and shrimp. NH4+ also substitutes for potassium (K+) in ion transporters, affecting the electrochemical gradient of the central nervous system (Cooper and Plum, 1987) and impairing brain function (Ip and Chew, 2010).
NO3– has low acute toxicity but can have significant chronic effects. NO3– can cause oxidative stress, which, if chronic, can weaken the immune system, affect reproduction, and slow growth. Its toxicity depends on concentration and exposure time (Pierce et al., 1993; Scott and Crunkilton, 2000), and also on salinity (Dowden and Bennett, 1965).
b. Indirect impacts of nitrogen emissions
When nitrogen discharge into water is excessive, nitrifying bacteria can be overwhelmed by high concentrations of NH3 and NO2–, causing them to accumulate in the water. Excess nitrogen stimulates the growth and proliferation of microalgae, leading to algal blooms. The high concentration of microalgae reduces light penetration in the water column, causing them to die. The decomposition of this organic matter by bacteria consumes large amounts of O₂, reducing its concentration in the water (Huo et al., 2013; Moss et al., 2013). This phenomenon is called eutrophication (Figure 4).
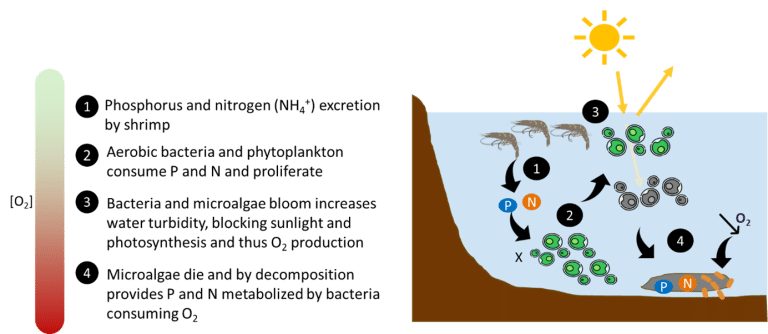
Figure 4: The process of eutrophication (Yang et al., 2008)
Eutrophication initiates a vicious cycle, with the lack of O2 in the water causing the death of other aquatic organisms, generating more organic matter and further reducing the O2 concentration (Yang et al., 2008). This has a serious impact on the growth and survival of fish and shrimp, particularly in ponds, which are closed systems with little water renewal. In particular, it has been observed that algae blooms in shrimp ponds in Mexico, caused by excessive nutrient input, have led to a 79.5% mortality rate among post-larvae, in addition to reducing growth (Licea, 1999).
In addition, eutrophication can lead to the proliferation of harmful species such as cyanobacteria, which often have an anaerobic metabolism, meaning that the decrease in oxygen concentration in the water promotes their proliferation (Codd, 2000). A decline in the survival of penaeid shrimp has been observed in China and Ecuador due to microalgae blooms causing anoxia in ponds (Alonso-Rodríguez and Páez-Osuna, 2003). In open systems such as sea cages, excessive nitrogen discharges can promote the proliferation of microalgae, some of which produce toxins (Anderson et al., 2002) or damage fish gills with their spicules (calcareous or siliceous spikes), potentially leading to significant mortality in affected fish farms (Burridge et al., 2010).
Recirculated aquaculture systems (RAS), thanks to their ability to control water parameters, are less affected by these issues. However, nitrogen discharge remains a significant problem even for the most technologically advanced systems. RAS filtration systems are very large and expensive to install. Reducing nitrogen levels in the water can help avoid oversizing filtration systems and thus limit costs. In addition, nitrification consumes the alkalinity of the water, requiring regular pH adjustments (Timmons et al., 2018).
4. The benefits of reducing nitrogen input in aquaculture systems
It is essential to control nitrogen levels, both in closed aquaculture facilities (such as tanks, ponds, and recirculation systems) to ensure animal performance and welfare, and in open systems (such as sea cages and raceways) to minimize their environmental impact. This can be achieved by reducing nitrogen emissions into the water or by improving the ability of nitrifying bacteria to convert nitrogen. Reducing nitrogen emissions involves optimizing feed distribution to avoid overfeeding and adjusting feed formulation to limit nitrogen intake. By precisely modulating the protein levels in feed in relation to other nutrients, protein catabolism and ammonia (NH3) emissions into water can be reduced.
This also involves choosing other raw materials, such as inorganic feed phosphates. It is crucial to choose products that limit phosphorus (P) and N emissions to prevent their accumulation in water and thus prevent eutrophication. For this purpose, monosodium phosphate (MSP) is the best choice, as it provides highly digestible P for fish and shrimp, while reducing N emissions into the water compared to monoammonium phosphate (MAP) (Morales et al., 2018). Although MAP phosphate also provides highly digestible P, it also provides excess NH3 that is not used by aquatic organisms in the same way as it is by ruminants.
Thus, while MAP is an effective phosphate for aquaculture, it increases nitrogen emissions into the water (Figure 5). Therefore, to provide an effective source of P while reducing P and N emissions, which affect fish health and the environment, it is preferable to opt for MSP.
4,2kg
This is the reduction in dissolved N excreted into the water per tonne of fish produced using MSP compared to MAP (Morales et al., 2018).
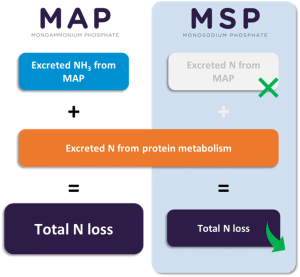
Figure 5: Diminution des rejets de N avec le MSP
Conclusion
Nitrogen is an essential element in aquaculture, but its metabolism can generate toxic waste such as NH3 when accumulated in water. Therefore, dietary adjustments to reduce N emissions and the creation of optimal conditions for nitrifying bacteria are necessary to minimize the harmful effects of N and ensure the health of aquaculture species. In terms of feeding and inorganic P intake, MSP is the most suitable source for ensuring the sustainability of aquaculture farms.
Bibliography
Alonso-Rodrı́guez, R., Páez-Osuna, F., 2003. Nutrients, phytoplankton and harmful algal blooms in shrimp ponds: a review with special reference to the situation in the Gulf of California. Aquaculture 219, 317–336. https://doi.org/10.1016/S0044-8486(02)00509-4
Anderson, D.M., Glibert, P.M., Burkholder, J.M., 2002. Harmful algal blooms and eutrophication: Nutrient sources, composition, and consequences. Estuaries 25, 704–726. https://doi.org/10.1007/BF02804901
Applegate, T.J., Angel, R., 2014. Nutrient requirements of poultry publication: History and need for an update. J. Appl. Poult. Res. 23, 567–575. https://doi.org/10.3382/japr.2014-00980
Beamish, F.W.H., Tandler, A., 1990. Ambient ammonia, diet and growth in lake trout. Aquat. Toxicol. 17, 155–166. https://doi.org/10.1016/0166-445X(90)90028-N
Bouwman, A.F., Pawłowski, M., Liu, C., Beusen, A.H.W., Shumway, S.E., Glibert, P.M., Overbeek, C.C., 2011. Global Hindcasts and Future Projections of Coastal Nitrogen and Phosphorus Loads Due to Shellfish and Seaweed Aquaculture. Rev. Fish. Sci. 19, 331–357. https://doi.org/10.1080/10641262.2011.603849
Brinkman, S.F., Woodling, J.D., Vajda, A.M., Norris, D.O., 2009. Chronic Toxicity of Ammonia to Early Life Stage Rainbow Trout. Trans. Am. Fish. Soc. 138, 433–440. https://doi.org/10.1577/T07-224.1
Bröer, S., 2008. Amino Acid Transport Across Mammalian Intestinal and Renal Epithelia. Physiol. Rev. 88, 249–286. https://doi.org/10.1152/physrev.00018.2006
Burridge, L., Weis, J.S., Cabello, F., Pizarro, J., Bostick, K., 2010. Chemical use in salmon aquaculture: A review of current practices and possible environmental effects. Aquaculture 306, 7–23. https://doi.org/10.1016/j.aquaculture.2010.05.020
Chang, Z.-W., Chiang, P.-C., Cheng, W., Chang, C.-C., 2015. Impact of ammonia exposure on coagulation in white shrimp, Litopenaeus vannamei. Ecotoxicol. Environ. Saf. 118, 98–102. https://doi.org/10.1016/j.ecoenv.2015.04.019
Ciji, A., Akhtar, M.S., 2020. Nitrite implications and its management strategies in aquaculture: a review. Rev. Aquac. 12, 878–908. https://doi.org/10.1111/raq.12354
Codd, G.A., 2000. Cyanobacterial toxins, the perception of water quality, and the prioritisation of eutrophication control. Ecol. Eng. 16, 51–60. https://doi.org/10.1016/S0925-8574(00)00089-6
Colt, J., Ludwig, R., Tchobanoglous, G., Cech, J.J., 1981. The effects of nitrite on the short-term growth and survival of channel catfish, Ictalurus punctatus. Aquaculture 24, 111–122. https://doi.org/10.1016/0044-8486(81)90048-X
Cooper, A.J., Plum, F., 1987. Biochemistry and physiology of brain ammonia. Physiol. Rev. 67, 440–519. https://doi.org/10.1152/physrev.1987.67.2.440
Cui, Y., Ren, X., Li, J., Zhai, Q., Feng, Y., Xu, Y., Ma, L., 2017. Effects of ammonia-N stress on metabolic and immune function via the neuroendocrine system in Litopenaeus vannamei. Fish Shellfish Immunol. 64, 270–275. https://doi.org/10.1016/j.fsi.2017.03.028
Dowden, B.F., Bennett, H.J., 1965. Toxicity of Selected Chemicals to Certain Animals. J. Water Pollut. Control Fed. 37, 1308–1316.
Engrola, S., Mai, M., Dinis, M.T., Conceição, L.E.C., 2009. Co-feeding of inert diet from mouth opening does not impair protein utilization by Senegalese sole (Solea senegalensis) larvae. Aquaculture 287, 185–190. https://doi.org/10.1016/j.aquaculture.2008.10.036
Evans, D.H., Claiborne, J.B., Currie, S., 2013. The Physiology of Fishes, Fourth Edition. CRC Press.
Forster, R.P., Goldstein, L., 1969. 5 Formation of Excretory Products, in: Hoar, W.S., Randall, D.J. (Eds.), Fish Physiology, Excretion, Ionic Regulation, and Metabolism. Academic Press, pp. 313–350. https://doi.org/10.1016/S1546-5098(08)60086-2
Frances, J., Allan, G.L., Nowak, B.F., 1998. The effects of nitrite on the short-term growth of silver perch (Bidyanus bidyanus). Aquaculture 163, 63–72. https://doi.org/10.1016/S0044-8486(98)00219-1
Gamboa-Delgado, J., Peña-Rodríguez, A., Ricque-Marie, D., Cruz-Suárez, L.E., 2011. Assessment of Nutrient Allocation and Metabolic Turnover Rate in Pacific White Shrimp Litopenaeus vannamei Co-Fed Live Macroalgae Ulva clathrata and Inert Feed: Dual Stable Isotope Analysis. J. Shellfish Res. 30, 969–978. https://doi.org/10.2983/035.030.0340
Han, S., Wang, B., Wang, M., Liu, Q., Zhao, W., Wang, L., 2017. Effects of ammonia and nitrite accumulation on the survival and growth performance of white shrimp Litopenaeus vannamei. Invertebr. Surviv. J. 14, 221–232. https://doi.org/10.25431/1824-307X/isj.v14i1.221-232
Hong, M., Chen, L., Sun, X., Gu, S., Zhang, L., Chen, Y., 2007. Metabolic and immune responses in Chinese mitten-handed crab (Eriocheir sinensis) juveniles exposed to elevated ambient ammonia. Comp. Biochem. Physiol. Part C Toxicol. Pharmacol. 145, 363–369. https://doi.org/10.1016/j.cbpc.2007.01.003
Huo, S., Ma, C., Xi, B., Su, J., Zan, F., Ji, D., He, Z., 2013. Establishing eutrophication assessment standards for four lake regions, China. J. Environ. Sci. 25, 2014–2022. https://doi.org/10.1016/S1001-0742(12)60250-2
Ip, A.Y.K., Chew, S.F., 2010. Ammonia Production, Excretion, Toxicity, and Defense in Fish: A Review. Front. Physiol. 1. https://doi.org/10.3389/fphys.2010.00134
Jensen, F.B., 2003. Nitrite disrupts multiple physiological functions in aquatic animals. Comp. Biochem. Physiol. A. Mol. Integr. Physiol. 135, 9–24. https://doi.org/10.1016/s1095-6433(02)00323-9
Kaushik, S.J., 1998. Whole body amino acid composition of European seabass (Dicentrarchus labrax), gilthead seabream (Sparus aurata) and turbot (Psetta maxima) with an estimation of their IAA requirement profiles. Aquat. Living Resour. 11, 355–358. https://doi.org/10.1016/S0990-7440(98)80007-7
Klocke, R., Andersson, K., Rotman, H., Forster, R., 1972. Permeability of human erythrocytes to ammonia and weak acids. Am. J. Physiol.-Leg. Content 222, 1004–1013. https://doi.org/10.1152/ajplegacy.1972.222.4.1004
Koo, J.-G., Kim, S.-G., Jee, J.-H., Kim, J.-M., Bai, S.C., Kang, J.-C., 2005. Effects of ammonia and nitrite on survival, growth and moulting in juvenile tiger crab, Orithyia sinica (Linnaeus). Aquac. Res. 36, 79–85. https://doi.org/10.1111/j.1365-2109.2004.01187.x
Licea, S., 1999. Proliferaciones de microalgas nocivas en los estanques para el cultivo semi-intensivo de camarón en México. Rev. Latinoam. Microbiol. 38, 123.
Lin, W., Luo, H., Wu, J., Hung, T.-C., Cao, B., Liu, X., Yang, J., Yang, P., 2023. A Review of the Emerging Risks of Acute Ammonia Nitrogen Toxicity to Aquatic Decapod Crustaceans. Water 15, 27. https://doi.org/10.3390/w15010027
Mallasen, M., Valenti, W.C., 2006. Effect of nitrite on larval development of giant river prawn Macrobrachium rosenbergii. Aquaculture 261, 1292–1298. https://doi.org/10.1016/j.aquaculture.2006.07.048
Molins-Legua, C., Meseguer-Lloret, S., Moliner-Martinez, Y., Campíns-Falcó, P., 2006. A guide for selecting the most appropriate method for ammonium determination in water analysis. TrAC Trends Anal. Chem. 25, 282–290. https://doi.org/10.1016/j.trac.2005.12.002
Morales, G.A., Azcuy, R.L., Casaretto, M.E., Márquez, L., Hernández, A.J., Gómez, F., Koppe, W., Mereu, A., 2018. Effect of different inorganic phosphorus sources on growth performance, digestibility, retention efficiency and discharge of nutrients in rainbow trout (Oncorhynchus mykiss). Aquaculture 495, 568–574. https://doi.org/10.1016/j.aquaculture.2018.06.036
Moss, B., Jeppesen, E., Søndergaard, M., Lauridsen, T.L., Liu, Z., 2013. Nitrogen, macrophytes, shallow lakes and nutrient limitation: resolution of a current controversy? Hydrobiologia 710, 3–21. https://doi.org/10.1007/s10750-012-1033-0
Nutrient Requirements of Fish and Shrimp, 2011. . National Academies Press, Washington, D.C. https://doi.org/10.17226/13039
Pierce, R.H., Weeks, J.M., Prappas, J.M., 1993. Nitrate Toxicity to Five Species of Marine Fish. J. World Aquac. Soc. 24, 105–107. https://doi.org/10.1111/j.1749-7345.1993.tb00156.x
Pinho, S.M., Emerenciano, M.G.C., 2021. Sensorial attributes and growth performance of whiteleg shrimp (Litopenaeus vannamei) cultured in biofloc technology with varying water salinity and dietary protein content. Aquaculture 540, 736727. https://doi.org/10.1016/j.aquaculture.2021.736727
Randall, D.J., Wright, P.A., 1987. Ammonia distribution and excretion in fish. Fish Physiol. Biochem. 3, 107–120. https://doi.org/10.1007/BF02180412
Ribeiro-de-Campos, B., Ortiz-Kracizy, R., Furtado, P.S., Volkweis-Zadinelo, I., Cupertino-Ballester, E.L., Ribeiro-de-Campos, B., Ortiz-Kracizy, R., Furtado, P.S., Volkweis-Zadinelo, I., Cupertino-Ballester, E.L., 2021. Effects of ammonia and nitrite on food consumption of the Amazon River prawn Macrobrachium amazonicum (Heller, 1862) postlarvae. Lat. Am. J. Aquat. Res. 49, 684–688. https://doi.org/10.3856/vol49-issue4-fulltext-2583
Robles-Porchas, G.R., Gollas-Galván, T., Martínez-Porchas, M., Martínez-Cordova, L.R., Miranda-Baeza, A., Vargas-Albores, F., 2020. The nitrification process for nitrogen removal in biofloc system aquaculture. Rev. Aquac. 12, 2228–2249. https://doi.org/10.1111/raq.12431
Rodríguez-Ramos, T., Espinosa, G., Hernández-López, J., Gollas-Galván, T., Marrero, J., Borrell, Y., Alonso, M.E., Bécquer, U., Alonso, M., 2008. Effects of Echerichia coli lipopolysaccharides and dissolved ammonia on immune response in southern white shrimp Litopenaeus schmitti. Aquaculture 274, 118–125. https://doi.org/10.1016/j.aquaculture.2007.10.049
Romano, N., Zeng, C., 2013. Toxic Effects of Ammonia, Nitrite, and Nitrate to Decapod Crustaceans: A Review on Factors Influencing their Toxicity, Physiological Consequences, and Coping Mechanisms. Rev. Fish. Sci. 21, 1–21. https://doi.org/10.1080/10641262.2012.753404
Roques, J., Schram, E., Spanings, T., 2013. The impact of elevated water nitrite concentration on physiology, growth and feed intake of African catfish Clarias gariepinus (Burchell 1822) – Roques – 2015 – Aquaculture Research – Wiley Online Library [WWW Document]. URL https://onlinelibrary.wiley.com/doi/abs/10.1111/are.12292 (accessed 10.29.25).
Scott, G., Crunkilton, R.L., 2000. Acute and chronic toxicity of nitrate to fathead minnows (Pimephales promelas), ceriodaphnia dubia, and Daphnia magna. Environ. Toxicol. Chem. 19, 2918–2922. https://doi.org/10.1002/etc.5620191211
Siikavuopio, S.I., Sæther, B.-S., 2006. Effects of chronic nitrite exposure on growth in juvenile Atlantic cod, Gadus morhua. Aquaculture 255, 351–356. https://doi.org/10.1016/j.aquaculture.2005.11.058
Svobodová, Z., Máchová, J., Poleszczuk, G., Hůda, J., Hamáčková, J., Kroupová, H., 2005. Nitrite Poisoning of Fish in Aquaculture Facilities with Water-recirculating Systems. Acta Vet. Brno 74, 129–137. https://doi.org/10.2754/avb200574010129
Thurston, R.V., Russo, R.C., 1983. Acute Toxicity of Ammonia to Rainbow Trout. Trans. Am. Fish. Soc. 112, 696–704. https://doi.org/10.1577/1548-8659(1983)112<696:ATOATR>2.0.CO;2
Timmons, M., Guerdat, T., Vinci, B., 2018. Recirculating Aquaculture, 4th edition.
Tomasso, J.R., 1994. Toxicity of nitrogenous wastes to aquaculture animals. Rev. Fish. Sci. 2, 291–314. https://doi.org/10.1080/10641269409388560
Weber, J.-M., Haman, F., 1996. Pathways for metabolic fuels and oxygen in high performance fish. Comp. Biochem. Physiol. A Physiol. 113, 33–38. https://doi.org/10.1016/0300-9629(95)02063-2
Weerd, J.H.V., 1995. Nutrition and growth in Clarias species – a review. Aquat. Living Resour. 8, 395–401. https://doi.org/10.1051/alr:1995046
Weihrauch, D., O’Donnell, M.J., 2015. Links between Osmoregulation and Nitrogen-Excretion in Insects and Crustaceans. Integr. Comp. Biol. 55, 816–829. https://doi.org/10.1093/icb/icv013
Yang, X., Wu, X., Hao, H., He, Z., 2008. Mechanisms and assessment of water eutrophication. J. Zhejiang Univ. Sci. B 9, 197–209. https://doi.org/10.1631/jzus.B0710626
