Introduction
Ruminants are different from other animals in view of their digestive system: they have a rumen. The rumen is the most important player of the digestion for ruminants, it is the place of biological fermentation by microorganisms. Then, feeding a ruminant is feeding its ruminal bacteria. Originally, ruminants are adapted to digest mainly forages but the evolution of production systems has transformed their diets. World milk production increased by more than 142 million tons of milk in ten years (+ 21.7 % ; FAO, 2024). To increase milk production or fattening performances, farmers feed more intensive diets, that if not well managed, disrupt ruminal environment and lead to Sub-Acute Ruminal Acidosis (SARA). SARA is a very common problematic in ruminants, as 19 % to 26 % of the cows in intensive dairy herds are affected by SARA (Kleen et al. 2013). The occurrence of SARA is associated with substantial impairments in animal health and performance, including 10% decline in fiber digestibility (Orne Conseil Elevage, 2016), and an average reduction of 2.7 kg of milk production per cow per day (Enemark, 2008). This expert file will go through SARA consequences and management.
What is subacute ruminal acidosis?
Subacute ruminal acidosis (SARA) is one of the main nutritional problems in dairy cows. This metabolic disorder results in a rumen fermentation deviation caused by an inadequate feed transition and/or diet composition. Resulting from an imbalance of the ruminal microflora caused by a diet rich in high fermentable carbohydrates and/or poor in proteins, SARA is a permanent threat for ruminants with a high level of production.
The optimal rumen pH for high producing dairy cows is between 6-6.5 which gives a balance of ruminal microflora. However, because of these new feed managements, daily fluctuations of ruminal pH are observed, irreversibly inducing a degradation of the ruminal papillae which leads to a decrease of the absorption capacity of nutrients (Figure 1). Therefore, these daily fluctuations of ruminal pH degrade health and zootechnical performances of animals.
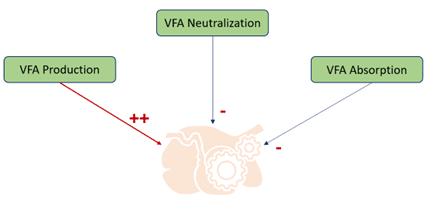
Figure 1: Origin of SARA
Rumen acidosis can be defined as a drop in the pH of rumen fluid following the overproduction of acids, particularly Volatile Fatty Acids (VFA) compared to neutralization and absorption. There are two forms of rumen acidosis: acute acidosis and subacute acidosis (Sauvant, Reverdin, Meschy 2006). It is the latter that we are going to develop.
SARA is the consequence of an energy and protein imbalance in the diet
The microorganisms of the rumen are very sensitive to the daily fluctuations of pH. To optimize the performances with an energy densification of the diet, cows are permanently on the razor edge of SARA. According to Plaizier et al. (2008), SARA can be described by an average ruminal pH of less than 5.6, for more than 3.5 hours per day. Indeed, when ruminal pH drops below 6, cellulolytic activity decreases and, on the other hand, amylolytic activity increases (Figure 2). The rumen no longer works properly: ruminal fermentations are reduced. As the cellulolytic bacteria allow to digest diet’s fiber, the digestion becomes incomplete. It is a vicious circle, because if this imbalance is not corrected quickly, the enzymatic activities of Lactobacillus (that prefer low pH) continue to increase, inducing a rise of lactate production and an important decrease of ruminal pH. Fermentations in the rumen continue to slow down until they stop completely, and the consequences (technical and economic) can be dramatic.
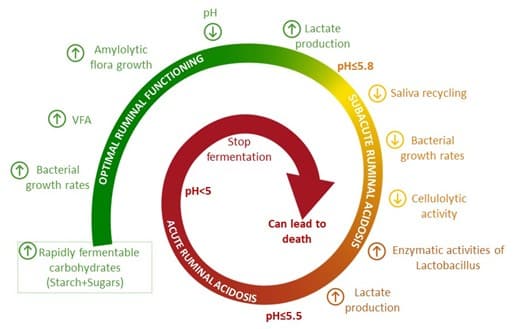
Figure 2: Vicious circle of acidosis. Adapted from Hernandez et al., 2014
Feed transition: a high-risk period for cows
Natural variations over time of corn or grass silos put diet’s stability through the mill. Indeed, corn silage continues to evolve during few weeks after its ensiling process. Corn silage derived from a single harvest can show week-to-week variations in its nutritional values (e.g. sugar content, digestibility of starch, dry matter content, …). However, with the low yields obtained in recent years, due to the drought, it is increasingly difficult to let the corn silage ferment for 1 to 3 months. That is why, cows are fed with fresh roughage which potentially induces digestive disorders. Compared to conserved roughage, fresh roughage does not have time to ferment, what increases the risk of SARA with the animal. Then, stability of the ruminal microflora is impacted. Indeed, fresh silage is rich in rapidly fermentable sugars. Once in the rumen, these sugars cause rapid and unbalanced fermentations. Amylolytic flora is over stimulated, producing high amounts of VFA, propionate and butyrate mainly. As explained earlier, this strong production of VFA exceeds absorption rate from ruminal wall and neutralization by saliva, increasing the risk of SARA.
Similar difficulties occur with spring and autumn grass. Indeed, young grass is very rich in rapidly fermentable carbohydrates (starch and sugars), in nitrogen and poor in crude fiber. Very palatable for animals, it is consumed quickly inducing less salivation and increasing the risk of SARA. Due to its high content in sodium bicarbonate (12.5 g/L according to Erdman, (1988)), saliva constitutes a natural buffer for ruminal pH. Thus, less saliva production means less rumen buffering.
High grain levels and starch source increase the risk of SARA
Diet composition is a key factor in SARA management. As an example, grain content but also grain origin can increase the risk of SARA. Elmhadi et al. (2022), affirm that a grain content between 50 % and 65 % can induce SARA. Khorrami et al. (2021), confirm that time with low pH (< 6) increases with the starch content in the diet. They also identified that type of starch used can impact the risk of SARA. Corn grain is more acidogenic than barley and wheat if fed in high quantity (> 20 % of the total dry matter ; Khorrami et al., 2021).
How to detect SARA?
Consequences of SARA can be numerous and can vary from one animal to another. They may take the form of one or more of the visual observations listed on Figure 3.
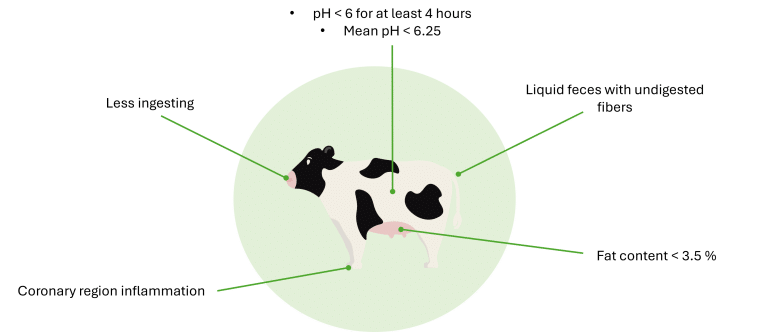
Figure 3: How to recognize SARA in animals?
By lowering dry matter intake (DMI ; -2.3 kg/day on average with SARA) and impacting rumen activity, SARA leads to milk production drop that can go from 1.8 kg/day to 3.3 kg/day according to Chiquette et al. (2015). Besides this production loss, the decreased activity of cellulolytic bacteria impacts milk fat content through lower production of acetate which is a VFA precursor of milk fat. Chiquette et al. (2015) observed a 0.18 % decrease in milk fat percentage during SARA challenge.
In young animals, SARA is also associated with reductions in DMI and average body weight gain in preweaning calves (Wenli Li et al., 2019).
Moreover, SARA can indirectly affect reproduction performances. Indeed, lower feed efficiency can lead to insufficient body condition that impacts the ability to conceive. As shown by Seesupa et al. (2017), SARA can also alter hormonal system resulting in lower fertility.
Considering all of this, SARA is costly for the farmer who can lose around 1.12-1.2€/cow/day (Plaizier et al., 2008).
How to limit the risk of SARA?
To reduce the risk of SARA, it is important to respect the basic principles of ruminant nutrition:
- Make progressive feed transitions
- Provide sufficient water
- Ensure a regular intake throughout the day
- Analyze the fodder to obtain a balanced diet in energy and protein
Diet profile in terms of starch and fiber is also very important. Especially fiber content and quality are key to manage the risk of SARA. As mentioned by Khorrami et al. (2021), cases of SARA increase when starch content is higher than 20 % and peNDF > 8 (peNDF is physically effective neutral detergent fiber, namely here, particle size bigger than 8 mm) is below 14 %. Nevertheless, peNDF must not be too high in order not to limit DMI.
Incorporating buffer solutions on a continuous basis or during periods of increased risk represents a key strategy to support animals and maintain optimal rumen activity in the context of SARA management. At Phosphea, we are committed to contribute to limit the risk of SARA by offering our natural buffer solution: CALSEA Powder.
How CALSEA Power can help with SARA?
CALSEA Powder is a natural solution 100% based on a calcareous marine algae (Lithothamnium calcareum). The porous structure of this calcareous red alga combined to its unique crystalline structure, allows a high solubilization and a fast release of calcium and carbonate ions in the rumen which enables a better maintain of ruminal pH.
Treatment with sodium bicarbonate (SBC ; 200 g/cow/day) has a lower mean ruminal pH than the treatment with CALSEA (100 g/cow/day ; Figure 4). The minimum ruminal pH obtained with treatment is 5.45 which is significantly lower than minimum ruminal pH obtained with CALSEA (Figure 4). Additionally, with CALSEA, the percentage of animals with a ruminal pH < 5.5 is significantly lower than with SBC (Figure 4). A pH < 6 for at least 4 hours, is a risk of acidosis and when pH<5.5, this risk is high. According to the results, CALSEA’s buffer capacity is higher than SBC’s one.
CALSEA Powder acts as a buffer solution that promotes ruminal pH stability, with subsequent positive effects on ruminal microbial populations and reduced risk of SARA. Figure 5 shows relative abundance of some bacteria that are indicators of SARA status with different buffer solutions.
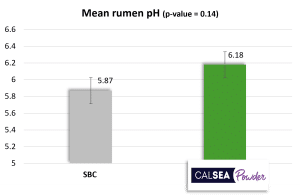
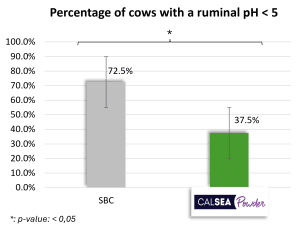
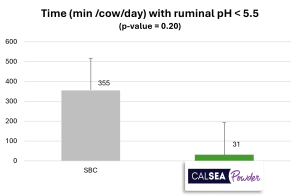
Figure 4: Action of CALSEA Powder on ruminal pH compared with sodium bicarbonate (in vivo trial ; n=40)
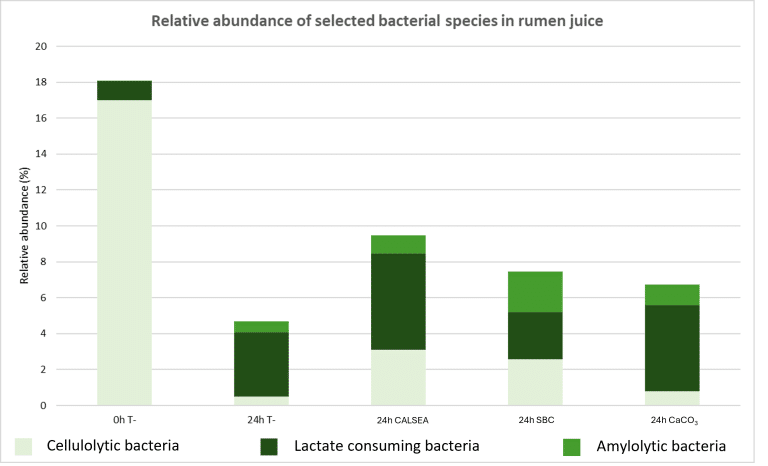
Figure 5: In vitro monitoring of ruminal bacterial populations in case of subacute ruminal acidosis (T- : Control with SARA)
0h T- represents the negative control without buffer at time 0. At this time, the acidogenic effect of the diet is not visible, the situation is considered normal. After 24 hours of fermentation without buffer (24h T-), SARA is installed and impacted negatively cellulolytic bacteria, their population dropped drastically. Compared with the negative control (24h T-), CALSEA (100 g/cow/day) and SBC (200 g/cow/day) appear to stimulate cellulolytic activity (Fibrobacter). Lactate consuming bacteria (Megasphaera) are more present with CALSEA and CaC03 than with SBC. This last bacterium protects against SARA because it consumes lactic acid. This trial shows that CALSEA promotes cellulolytic bacteria activity under acidogenic conditions, thereby helping to maintain optimal rumen efficiency.
Products tailored to your livestock's need
Bibliography
CHIQUETTE, J., LAGROST, J., GIRARD, C.L., TALBOT, G., LI, S., PLAIZIER, J.C. and HINDRICHSEN, I.K., 2015. Efficacy of the direct-fed microbial Enterococcus faecium alone or in combination with Saccharomyces cerevisiae or Lactococcus lactis during induced subacute ruminal acidosis. Journal of Dairy Science. January 2015. Vol. 98, n° 1, pp. 190‑203. DOI 10.3168/jds.2014-8219.
ELMHADI, Mawda E., ALI, Darien K., KHOGALI, Mawahib K. and WANG, Hongrong, 2022. Subacute ruminal acidosis in dairy herds: Microbiological and nutritional causes, consequences, and prevention strategies. Animal Nutrition. September 2022. Vol. 10, pp. 148‑155. DOI 10.1016/j.aninu.2021.12.008.
ENEMARK, Jörg M. D., 2008. The monitoring, prevention and treatment of sub-acute ruminal acidosis (SARA): A review. The Veterinary Journal. April 2008. Vol. 176, n° 1, pp. 32‑43. DOI 10.1016/j.tvjl.2007.12.021.
ERDMAN, Richard A., 1988. Dietary Buffering Requirements of the Lactating Dairy Cow: A Review1. Journal of Dairy Science. December 1988. Vol. 71, n° 12, pp. 3246‑3266. DOI 10.3168/jds.S0022-0302(88)79930-0.
FAOSTAT, [on line]. [Visited on January, 8th 2026]. Available on: https://www.fao.org/faostat/en/#data/QCL
KHORRAMI, Behzad, KHIAOSA-ARD, Ratchaneewan and ZEBELI, Qendrim, 2021. Models to predict the risk of subacute ruminal acidosis in dairy cows based on dietary and cow factors: A meta-analysis. Journal of Dairy Science. July 2021. Vol. 104, n° 7, pp. 7761‑7780. DOI 10.3168/jds.2020-19890.
KLEEN, J. L., HOOIJER, G. A., REHAGE, J. and NOORDHUIZEN, J. P. T. M., 2003. Subacute Ruminal Acidosis (SARA): a Review. Journal of Veterinary Medicine Series A. 2003. Vol. 50, n° 8, pp. 406‑414. DOI 10.1046/j.1439-0442.2003.00569.x.
LI, Wenli, GELSINGER, Sonia, EDWARDS, Andrea, RIEHLE, Christina and KOCH, Daniel, 2019. Transcriptome analysis of rumen epithelium and meta-transcriptome analysis of rumen epimural microbial community in young calves with feed induced acidosis. Scientific Reports. March 2019. Vol. 9, pp. 4744. DOI 10.1038/s41598-019-40375-2.
PLAIZIER, J. C., KRAUSE, D. O., GOZHO, G. N. and MCBRIDE, B. W., 2008. Subacute ruminal acidosis in dairy cows: The physiological causes, incidence and consequences. The Veterinary Journal. April 2008. Vol. 176, n° 1, pp. 21‑31. DOI 10.1016/j.tvjl.2007.12.016.
SAUVANT, D., REVERDIN, Stéphane et MESCHY, François, 2006. Le contrôle de l’acidose ruminale latente. INRA Productions Animales. 2006. Vol. 19, n° 2, pp. 69‑78
